Clinical Evidence
Independent evaluations examining Simini Protect Lavage under controlled intraoperative conditions.
The evidence summarized below focuses on measurable intraoperative activity at surgical closure and is intended to inform closure-stage decision-making.
How intraoperative lavage strategies are evaluated
Intraoperative lavage strategies are evaluated by measuring residual viable bacteria at the point of surgical closure, the final moment under direct surgical control.
Standardized activity-based models isolate the effect of a lavage intervention by controlling inoculum, exposure conditions, and recovery methods.

Endpoint: Residual viable bacteria (CFU) measured at closure
Comparator: Saline and other antiseptics under identical conditions
Models: In vitro, implant-associated, biofilm-associated, and in vivo surgical models
Role: Adjunctive lavage within standard orthopedic workflows
What different study designs evaluate
Different study designs answer different questions in orthopedic infection control.
Randomized clinical trials are designed to evaluate population-level postoperative outcomes, such as surgical site infection rates. These outcomes are influenced by host factors, surgical technique, environment, and postoperative care, making it difficult to isolate the contribution of a single intraoperative step.
Activity-based intraoperative studies are designed to measure what happens during surgery. By assessing residual bacterial burden at closure under standardized conditions, these models isolate the intraoperative activity of a lavage strategy itself.
These approaches are complementary and address different aspects of infection risk.
Findings across controlled orthopedic models
Across multiple independent evaluations, Simini Protect Lavage demonstrated lower recoverable bacterial burden at surgical closure compared with saline and other antiseptics when assessed under identical experimental conditions.1-5
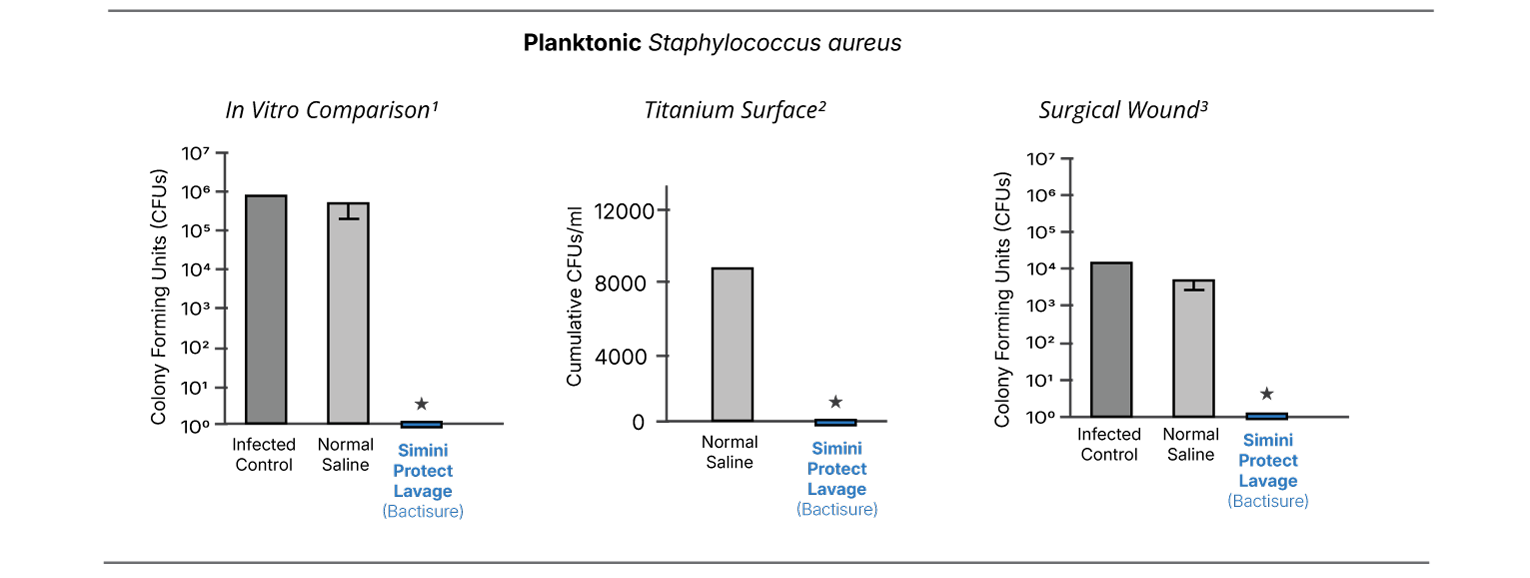
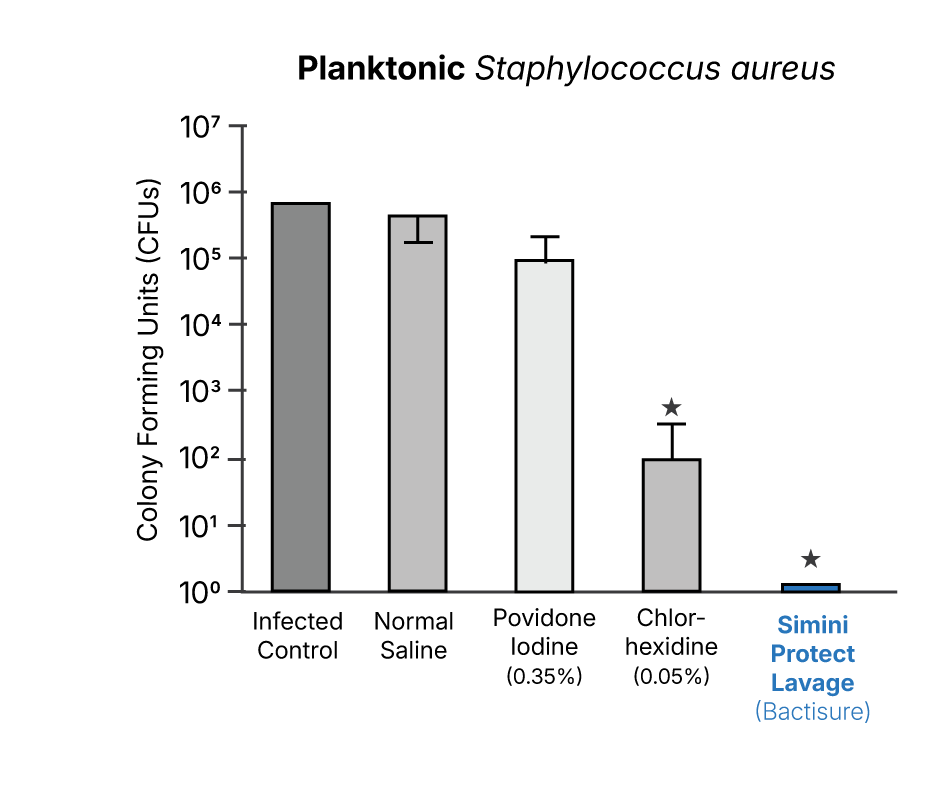
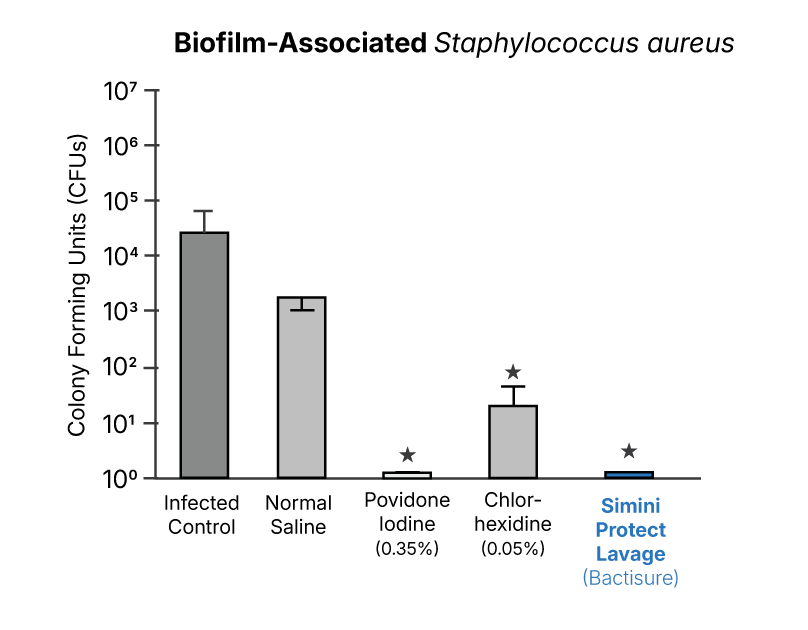
Biofilm-associated and revision surgery contexts
Biofilm-associated models evaluate bacterial adherence and persistence on implant materials under controlled laboratory and surgical conditions.
In addition to controlled experimental models, published clinical case series from veterinary orthopedic practice—including work conducted at Clinica Vezzoni (Cremona) and reported by Forzisi et al.—provide real-world context regarding use in complex surgical environments.
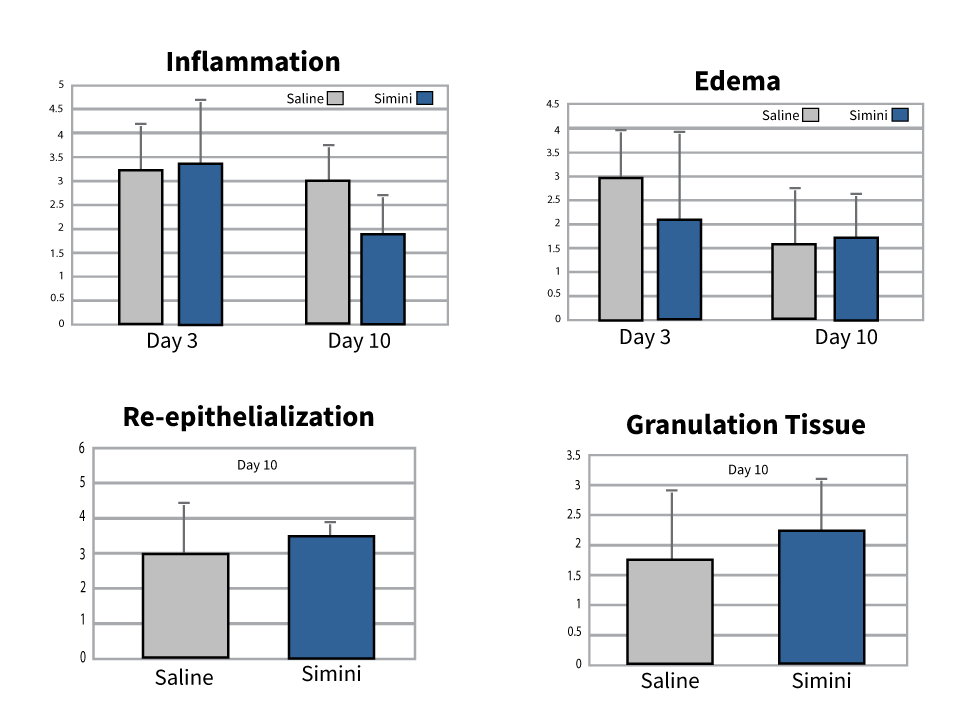
Safety and tissue compatibility under intraoperative conditions
- No delayed or impaired wound healing compared with saline when used as directed 9,11
- Compatibility has been evaluated in murine, porcine, and ISO implantation models 10
- Findings support use around orthopedic implants within routine closure workflows
Data resources
References
- Hamad C, Sheppard W, Chun R, Mamouei Z, Lloyd T, Li A, Sobti R, Pearce J, Bernthal N, McPherson E, "Comparing the in Vitro Efficacy of Commonly Used Surgical Irrigants for the Treatment of Implant-Associated Infections" The Journal of Bone and Joint Surgery (2025)
- Seta J, Pawlitz P, Aboona F, Weaver M, Bou-Akl T, Ren W, Markel D, "Efficacy of Commercially Available Irrigation Solutions on Removal of Staphylococcus Aureus and Biofilm From Porous Titanium Implants: An In Vitro Study" The Journal of Arthroplasty 39 (2024)
- Texas Tech Murine Surgical Model - Data on file
- O'Donnell J, Wu M, Cochrane N, Belay E, Myntti M, James G, Seyler T, "Efficacy of Common Antiseptic Solutions Against Clinically Relevant Planktonic Microorganisms," Orthopedics (2022)
- Kia C, Cusano A, Messina J, Muench LN, Chadayammuri V, McCarthy MB, Umejiego E, Mazzocca AD, "Effectiveness of topical adjuvants in reducing biofilm formation on orthopedic implants: an in vitro analysis", Journal of Shoulder and Elbow Surgery (2021)
- Forzisi I, Vezzoni L, Bozzerla M, Vezzoni A: "Use of Simini Protect Lavage as an Adjuvant in the Antiseptic Protocol for Revision Surgeries Involving Total Hip Replacement", Veterinary and Comparative Orthopaedics and Traumatology Open Vol. 8 No. 1 (2025)
- I. Forzisi, L. Vezzoni, M. Bozzerla, A.Vezzoni "Intraoperative Antiseptic Lavage with Simini for Prevention and for Treatment of SSI." European Society of Veterinary Orthopaedics and Traumatology Meeting, Sept 24, 2022
- Hunter et al., "Clinical Effectiveness of a Biofilm Disrupting Surgical Lavage in Reducing Bacterial Contamination in Total Knee Arthroplasty Revision Surgery in Known Cases of Prosthetic Joint Infection", Zimmer Biomet white paper. (administered 1 litre volume via pulsed jet lavage)
- Powell et al., "Effect of Commercially Available Wound Irrigation Solutions on Uninfected Host Tissue in a Murine Model", Arthroplasty Today 2024 Jan 13:25 (Bactisure and Simini Protect Lavage are the same fluid)
- ISO Intramuscular Implantation Test with Histopathology (14 Day Rabbit Study)- Simini Protect Lavage - No abnormal clinical signs, Lavage was considered "Non-Irritant" compared to predicate device.
- Dermal Testing - Uninfected wounds (14 Day Porcine Study) Simini Protect Lavage resulted in normal wound healing and was comparable to a predicate device.
 EN ▾
EN ▾ ES
ES IT
IT FR
FR DE
DE