Closure is the final controllable moment
in orthopedic surgery.
Simini Protect Lavage is a non-antibiotic intraoperative lavage solution with a physical mechanism independent of antibiotic activity, used as a final lavage step prior to closure to address residual bacterial burden in the surgical field.

Product Profile
A practical summary for surgeons evaluating Simini Protect Lavage.
- Residual viable bacteria (CFU) measured at closure
- Activity assessed using standardized orthopedic surgical models
- Non-antibiotic; compatible with standard antibiotic protocols
- Evaluated for use around orthopedic implants and healing tissues
Closure is where surgical control ends.
By the time skin is closed, implants are placed, tissue is exposed, and the surgical field is no longer accessible.
Decisions made at closure define the transition from surgical control to biological healing. This moment is consistent across routine and complex orthopedic procedures.

Residual bacterial burden is a measurable variable at closure.
Postoperative infection rates are influenced by host, environment, and follow-up variables.
Because postoperative infection is influenced by many variables, intraoperative strategies are often evaluated by measuring viable bacterial burden at the time of closure.
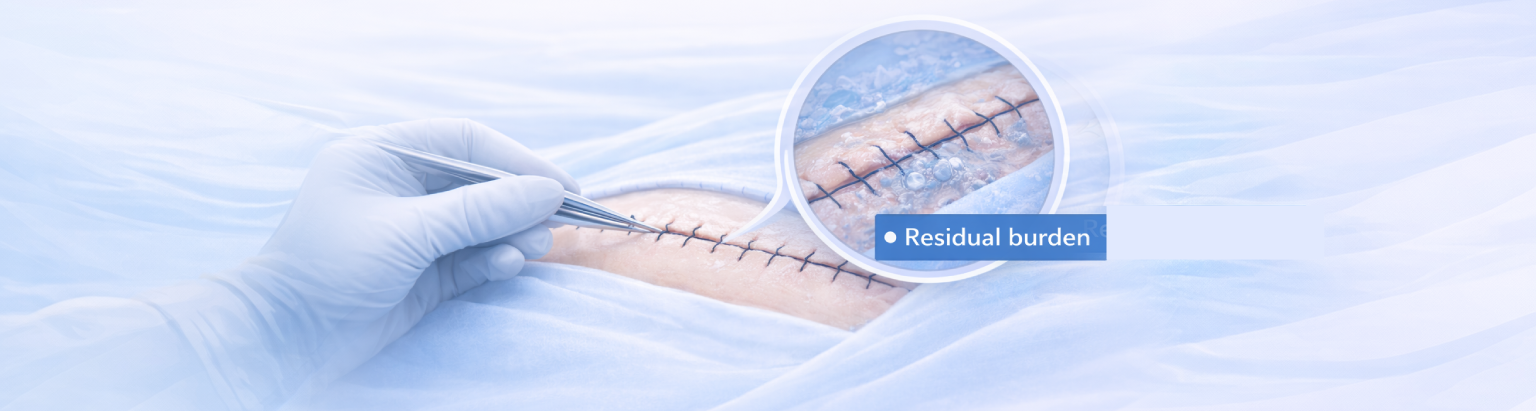
Closure is addressed through standardized protocols.
When closure is treated as a distinct phase of surgery, consistency becomes possible. Standardized closure protocols reduce variability at the final controllable moment, while fitting within existing orthopedic workflows.
TPLO is often the first procedure where these protocols are adopted due to frequency, implant use, and functional demand.

Studied within standardized orthopedic models.
Simini Protect Lavage has been studied using reproducible microbiologic methods measured at closure. These evaluations include routine orthopedic procedures, implant-associated models, and higher-challenge surgical environments. This evidence is intended to inform closure-stage decision-making, not replace clinical judgment.
Endpoint: Residual viable bacteria (CFU) measured at closure
Design: Controlled, standardized intraoperative evaluations
Findings: Simini demonstrated measurable reduction in residual bacterial burden at closure under standardized conditions
View detailed evidence summaries →
Designed to integrate without changing how you operate.
Simini Protect Lavage is used as a final adjunct prior to closure. It does not alter instrumentation, timing, or operative sequence, and fits within existing orthopedic workflows. Designed for orthopedic procedures where consistency at closure matters.



Adds approximately 1 minute as a final lavage step prior to closure.
Safety & compatibility
- Non-antibiotic; does not replace or interfere with systemic antibiotic protocols
- Evaluated for compatibility with commonly used orthopedic implant materials
- No known adverse effects on wound healing when used as directed
- Intended for orthopedic-associated exposure at closure
- Refer to the User Guide and Instructions for Use for contraindications and handling guidance
How closure is addressed is a clinical decision.